Reconstruction
Contents
Below we give an overview of matching defect types and sites with reconstructive options.
Some general considerations
Reconstructive surgery is based on the firm working principle that tumour ablation should never be compromised for the sake of reconstruction.
Some things to keep in mind:
- defect size, site and tissue character:
- flap characteristics (for example, tissue quality and quantity available, pedicle length, vascular reliability including vessel calibre), success rate, donor site morbidity and complications;
- functional outcome options available (for example, appearance, chewing, quality of life, sensation, shoulder function, speech, swallowing);
- rehabilitation options (for example, dental implants);
- cost factors and resource requirements;
- a patient’s general health (for example, comorbidities can influence reconstructive options);
- a patient’s preference;
- the surgeon’s preference (and expertise);
- alternative options (for example, non-surgical options such as an obturator, or no reconstruction).
The reconstructive ladder
With the many different reconstructive techniques available, one useful classification frequently quoted is the reconstructive ladder. This ladder starts at the bottom rung with the simplest technique, open wound healing, and progresses to prefabricated free flaps (or possibly composite tissue allotransplantation) at the top:
open wound healing => primary closure => graft => local flap => distant pedicled flap => distant free flap => prefabricated flaps.
This is a useful concept. It reflects the expertise involved and, when linked with consequences of failure, as in ‘snakes and ladders’, aptly reflects the ‘height of the fall’ should the technique not work. It is important to realise that although the more sophisticated technique often has greater benefits, it is likely that a higher price will be associated with its failure. For example, a free flap is usually an all or none phenomenon, and every graft or flap harvested has accompanying donor site morbidity.
This field of surgical activities may be the most in need of recalling Boyes’ law: ‘The ability to do an operation is not an indication’. An even more graphic version of this advice from surgeon to surgeon would be: ‘Keep it simple, stupid’.
Facial skin
Apart from open wound healing and primary closure, skin grafts as well as local flaps play an important role in the reconstruction of facial skin defects.
Repair of facial skin defects with local flaps often is a preferred option, where possible: local flaps provide excellent tissue match and very satisfactory cosmesis. Not all facial skin defects can be reconstructed with local flaps, and skin grafts (particularly full-thickness grafts) are an alternative solution.
Skin grafts may be the treatment of choice in certain defects, but equally they may be the only option because of a lack of adjacent tissue or the size of the defect. Three types of skin grafts are commonly used: full-thickness, split-thickness and composite grafts. The full-thickness skin graft is the most useful for facial reconstruction. Commonly used as the treatment of choice for the nasal tip and the lower eyelid, it is also useful for defects of the nasal alae (see Figure 1) and the ear, as well as for larger defects of the temple and forehead. Suitable donor sites are the postauricular (behind the ear) region and the supraclavicular (above the collarbone) area of the neck.
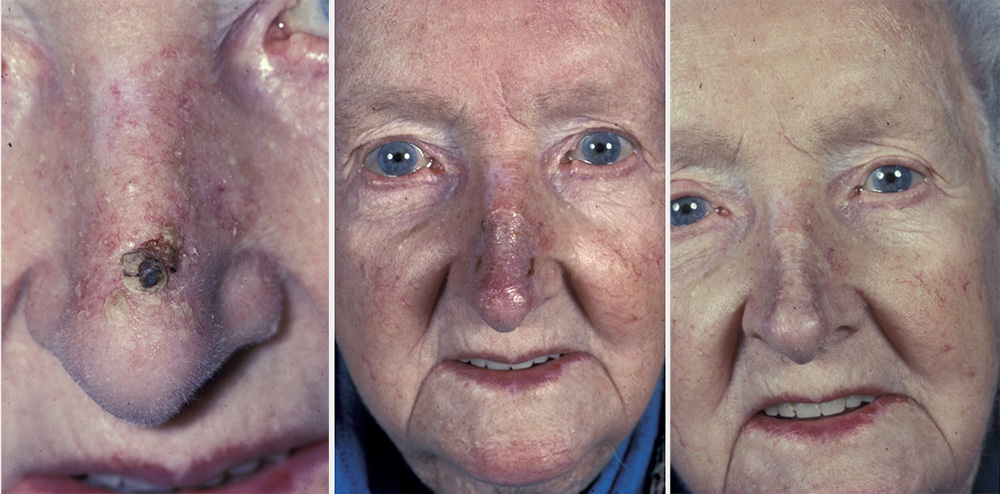
The perichondrial cutaneous graft is more substantial and three-dimensionally stable than normal full-thickness skin grafts. It is harvested from the conchal bowl of the ear by taking both skin and perichondrium for grafting of the nose or eyelids. This donor site defect is reconstructed with an island flap (‘flip flop flap’) from the postauricular sulcus.
Split-thickness grafts are harvested from the inner thigh (see Figure 2) or arm either manually or with an electric dermatome.
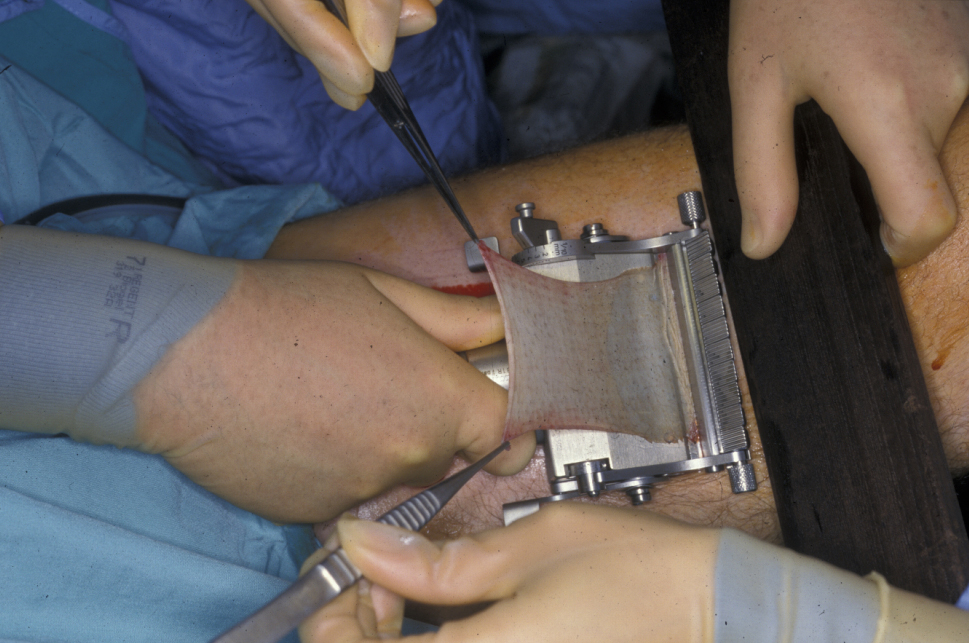
Split-thickness grafts are useful for large defects, particularly of the scalp (see Figure 3). Because these grafts allow more contraction than full-thickness grafts, they must be used with care on the face and close to free margins and are usually the last reconstructive choice in these areas.
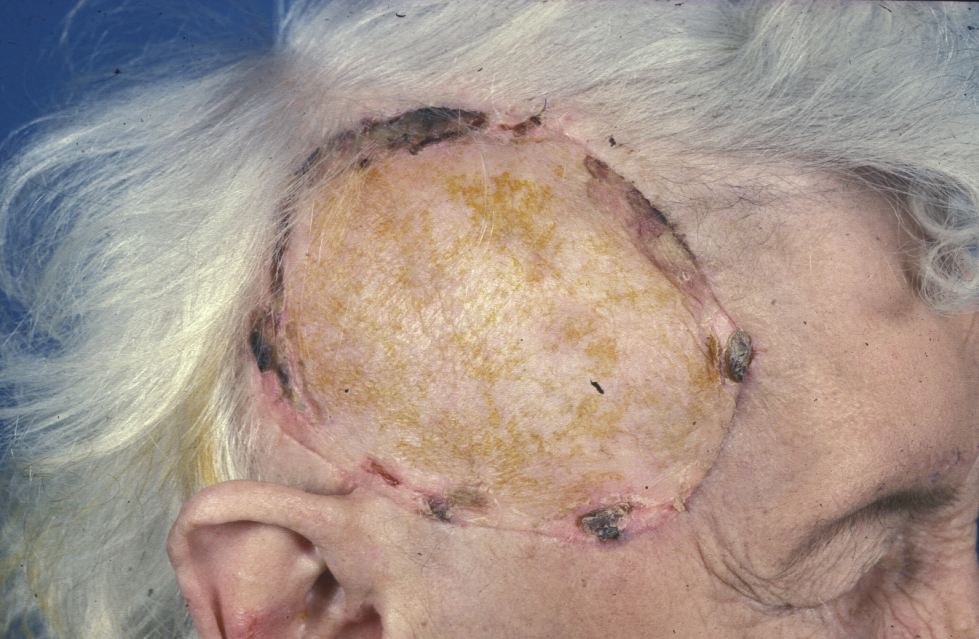
For all skin grafts there must be sufficient vascular tissue in the defect bed to support the graft, to allow initial diffusion of oxygen and nutrients to the graft (osmotic imbibition), followed by re-vascularization around the fourth to seventh day. Bare bone, tendon or cartilage will not allow grafts to take, and other reconstructive options must be employed. Following skin grafting to a defect site, the avoidance of movement or shear of the graft on the vascular bed during the healing phase is vital and necessitates careful stabilisation of the graft with sutures and appropriate dressings such as a tie-over pack (Figure 4) or negative-pressure splinting for up to 10 days. Infection and development of a haematoma between the graft and the defect bed are the two other major causes of graft failure,
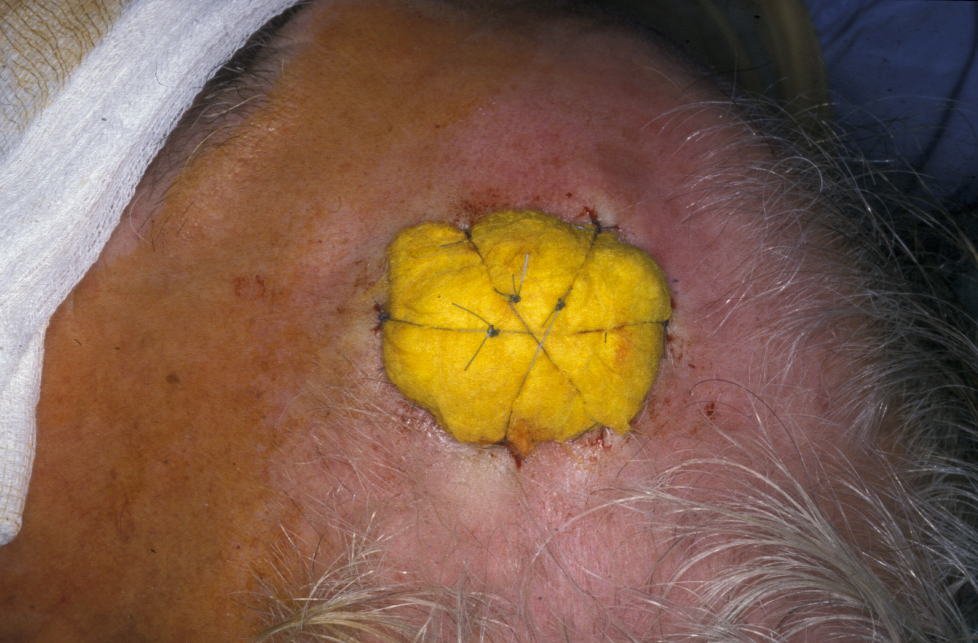
When properly chosen, skin grafts are often functionally and aesthetically successful. The disadvantages are that the colour, texture and physical bulk may not match the original tissue and may be inferior to the result that could be achieved with a local flap. Occasionally hypo- or hyperpigmentation can occur in the skin graft, and it is cosmetically detrimental.
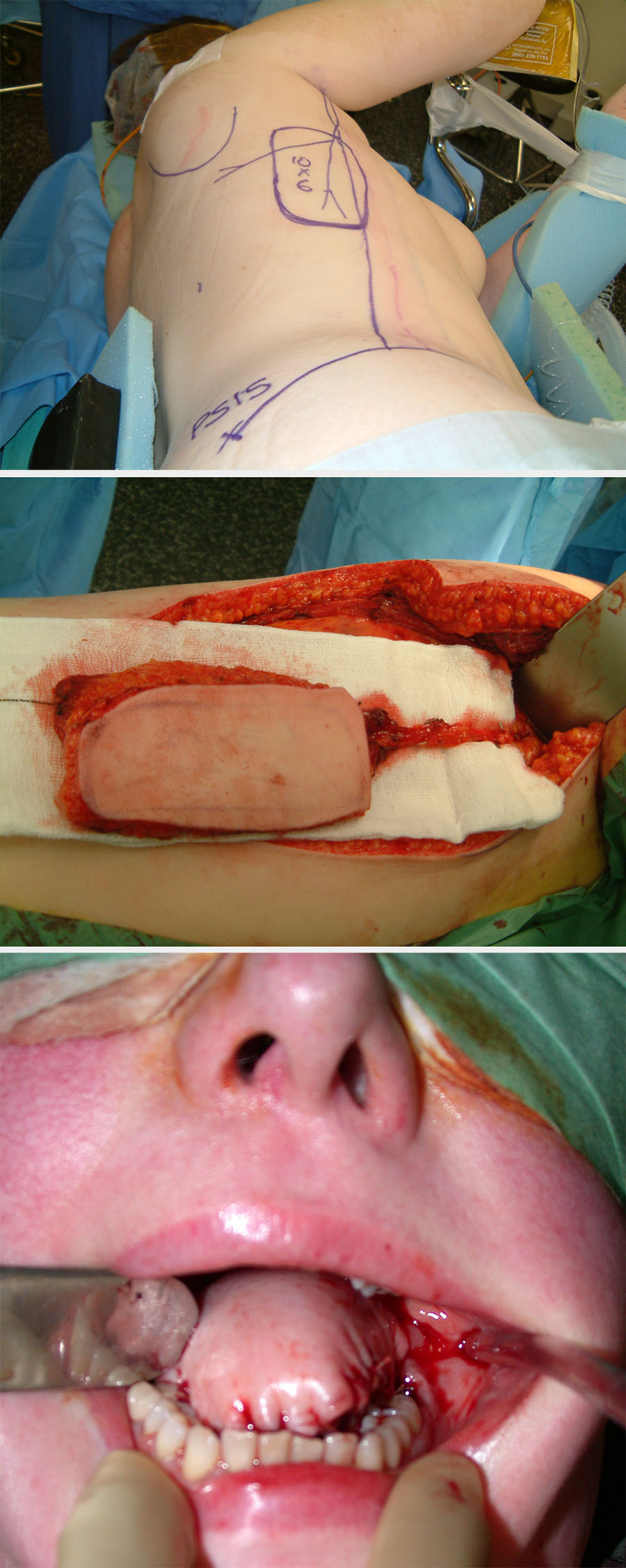
Ventral (underside) tongue and floor of the mouth
The main aim in reconstructing these defects is to allow maximal movements of the residual tongue so that the best possible speech, eating and swallowing can be achieved. Small lesions can be excised with a laser and left to heal or can be excised and repaired with a skin graft, or the wound sometimes can be primarily closed with sutures (longitudinal better than transverse to reduce tethering). Larger defects can be repaired with a local flap (for example, a nasolabial flap or a radial forearm free flap).
Where the majority of the tongue has been removed, there will be little or no residual function. This defect is best reconstructed with bulk (for example, a pedicled pectoralis major flap) or large free flaps such as a latissimus dorsi flap (see Figure 5), or a rectus abdominis flap.
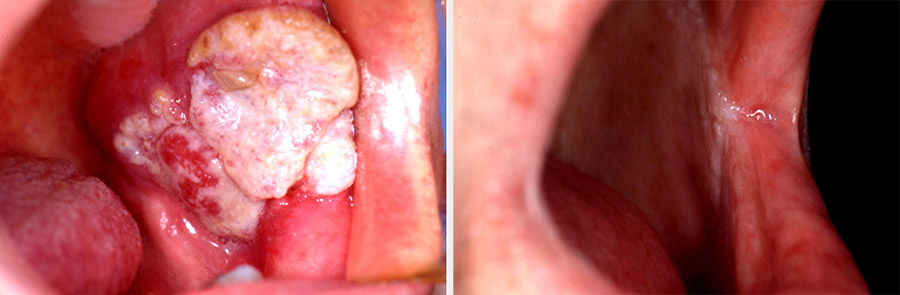
Buccal mucosa
The main requirement for this defect is to obtain closure by covering the area with a pliable lining while allowing for normal mouth opening. Superficial lesions can be excised with a laser and left to heal or repaired with a skin graft. Small deeper defects can be closed with a local flap. A large and deep defect is better repaired with a radial forearm free flap (see Figure 6). Through-and-through defects require bipaddled repair.
Mandible
Where a segmental resection is required for bone invasion by cancer (see Figure 7), it is likely that the area will receive radiotherapy as an adjuvant measure for local control. A free non-vascularized bone graft is unlikely to survive such hostile treatment. Ideally, a vascularized bone flap followed by dental implant insertion should be considered for optimal rehabilitation. Suitable flaps include the fibula (good length; see Figure 7), which can be combined with other flaps because of its long pedicle (see Figure 7), and the DCIA (deep circumflex iliac artery; large bone stock but much shorter pedicle; see Figure 8) free flaps.
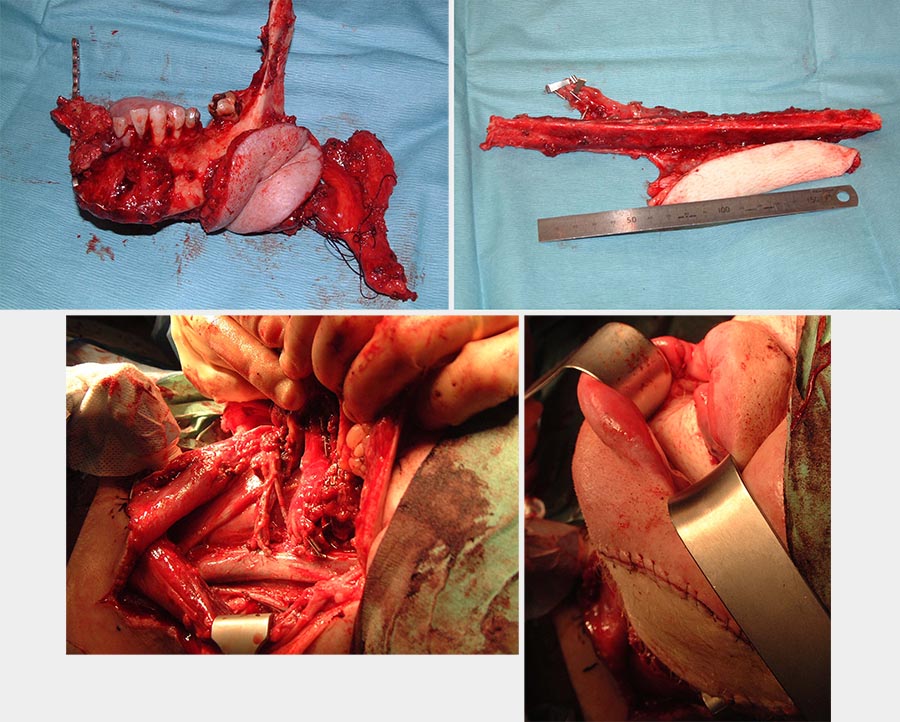
Anterior (front) segments of the mandible if left unreconstructed will leave severe deformity and collapse of the chin with drooling. There are few reasonable alternatives to composite free flaps. Lateral (side) defects of the mandible, although best restored by composite flaps, can be simply treated with a reconstruction plate to maintain continuity with an enveloping bulky soft tissue flaps (such as the pectoralis major flap) when somebody is not fit for a free-flap surgical procedure.
The available options for mandibular reconstruction include:
- Free bone grafts – a corticocancellous block graft is taken from the anterior (front) or posterior (back) iliac crest. Free bone grafts are still a good option for defects that are not bigger than approximately 5 cm, provided that the soft tissues are in good condition. Gaps larger than 5 cm are not really suitable for this means of bridging a defect. In most cases of malignancy when a large part of the surrounding soft tissues is lacking, healing cannot be ensured. Previous or anticipated radiotherapy would also be a contraindication for the use of free bone grafts. Studies comparing the anterior and posterior iliac crest as donor sites reveal less donor site morbidity when the posterior site is used.
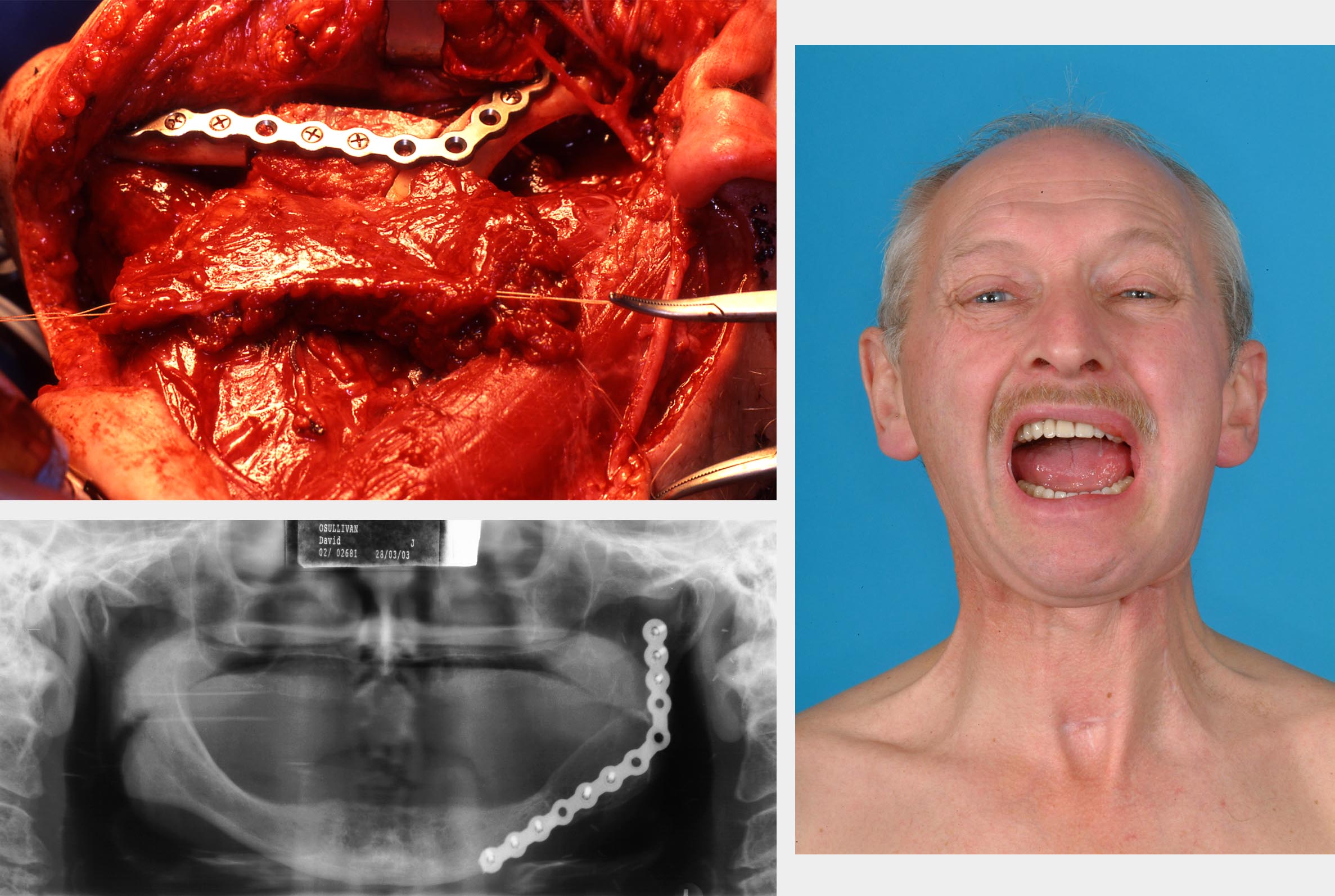
- Reconstruction plates – these are rigid plates that are applied along the lower border of the mandible. They were made with the intention of bridging a defect, thus will stabilise occlusion and facial contour. They are currently used to fix corticocancellous blocks or vascularized bone grafts to the remaining mandible. There are several types of these plates on the market, but the overriding principle is to have one single plate of sufficient thickness and width to hold the fragments in place, approximately 3 mm thick and 5 mm wide. A special feature is the locking screw. This type of screw is supposed to minimise compression between the plate and the underlying bone and thereby optimise the vascularity surrounding the graft.
- Free flaps – these include fibula, DCIA and scapula free flaps.
- Particulate cancellous bone and marrow grafts – these must be placed in a frame (a crib).
- Transport disc distraction osteogenesis – a segment of bone is cut adjacent to the defect (transport disc) and is moved gradually across the defect.
Maxilla
The main aims of rehabilitating this defect are to achieve coverage and to seal the oral cavity from the floor of nose and antrum. This has been traditionally managed with an obturator (prosthesis; see below). Where facial skin and the orbit can be preserved, it remains a reasonable option by allowing a patient to speak, eat and look normal while the obturator is worn.
There is a trend toward microvascular reconstruction of maxillary defects with the aim of doing away with the prosthesis. This technique has a better outcome for larger defects than obturation and is much preferable if facial skin is involved in the defect. It is technically more challenging, and care is required to ensure that adequate pedicle length and suitable vessels are available for anastomosing the flap. Flaps commonly used for this defect include DCIA, fibula, rectus abdominis, latissimus dorsi and radial forearm flaps (see Figure 9 and Figure 10 for examples).
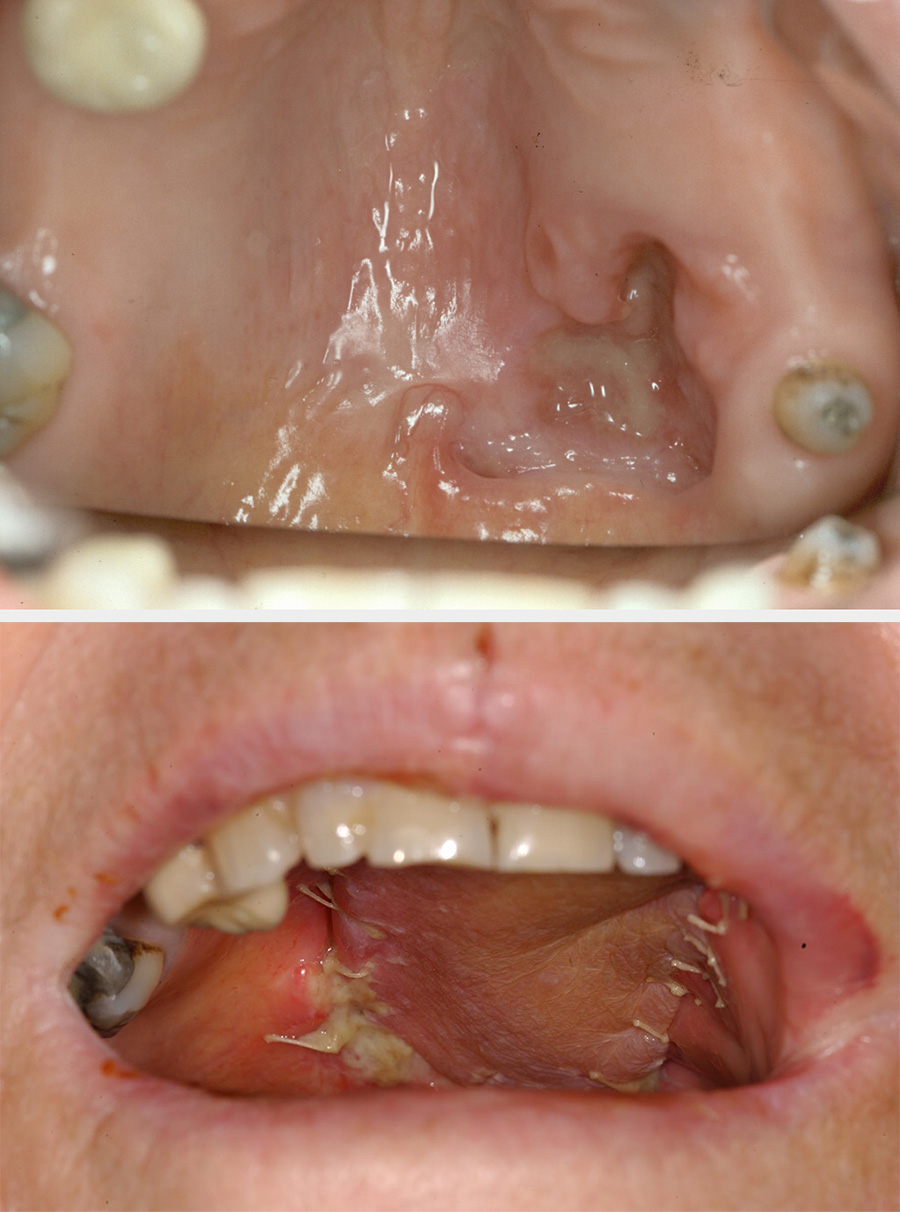
Lip
The lips are an important area in terms of cosmesis and function (lip seal, speech and eating). Full-thickness defects of up to one third of the lip can be primarily closed. Intermediate full-thickness defects can be reconstructed with local flaps, or Abbe flaps. An Abbe flap involves rotating a wedge of lip from the opposite side of the lip by using a two-staged technique. Total full-thickness lip defects can be repaired using bilateral fan flaps, or with a free tissue transfer with tendon slings.
Pharynx
An intact oropharynx is vital for swallowing and speech. Partial defects can be repaired using a rigid bulky flap such as the pectoralis major flap with the aim of allowing remaining tissue to have some function. If the defect requires the creation of a circumferential tube, alternatives include free soft tissue transfers such as a tubed radial forearm (largely historical now) or anterolateral thigh and jejunal free flaps.
Prosthetic rehabilitation
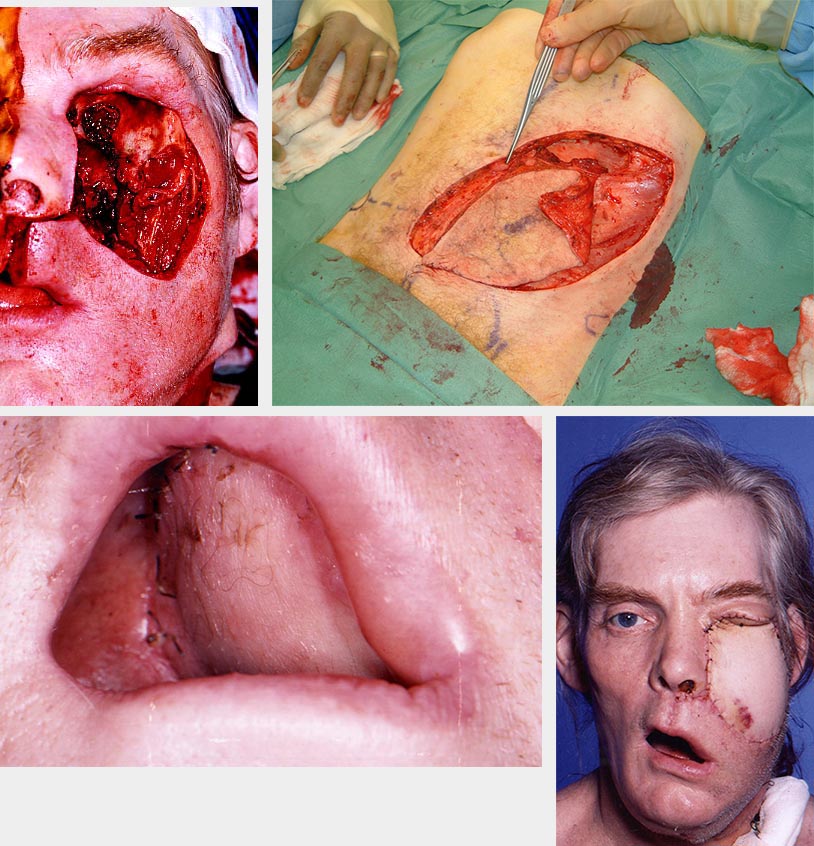
The use of nonbiological materials to reconstruct certain head and neck defects is an established and effective option. These defects include auricular (ear; implant-retained ear prosthesis), maxillary and total rhinectomy (nose resection) defects (prosthesis retained with undercuts or implants). It is important to assess somebody’s compliance and dexterity (to keep implants clean and to remove and reinsert prostheses) at the planning stage. This preoperative assessment and planning should be carried out with prosthodontist and maxillofacial technician so that optimal results can be achieved.
Maxillary prosthesis
This removable reconstructive option involves taking an impression before surgery to make a dressing plate. When the surgical defect is created, the dressing plate is used with a suitable obturator material (gutta percha or silicone) to provide support for the cheek, create an undercut to aid retention of the prosthesis and seal the oral cavity from the nasal and antral cavities to enable speech and swallowing. The plate can be secured with wires or screws. The inner wall of the defect is usually grafted with split-thickness skin to speed up healing, and coroniodectomy (removal of a small top part of the mandible) is performed to stop dislodgment of the obturator by mandibular lateral excursions.
The dressing plate and obturator are removed several weeks later for impressions to reconstruct an interim removable obturator. This is followed by a definitive prosthesis several months later. The prosthesis can be a single piece hollowed (to reduce weight and aid retention) or two-pieced (soft bung superiorly with rigid conventional oral plate) prosthesis. Existing teeth within the same arch can be used to improve retention of the prosthesis with the aid of clasps. The maxillary obturator prosthesis for low-level maxillary defects is an excellent reconstructive option – so much so that some patients tend to refuse any secondary flap options if offered.
Solitary ear prosthesis was traditionally retained with glue (unsatisfactorily!). The advent of osseointegrated implants heralded a major improvement in this area. Larger complex defects involving the skin can be ‘reconstructed’ using multi-pieced obturators retained with implants, interconnecting joints or spectacles. However, the skin component coloration tends to fade with time and sun exposure. Facial components look good when they are still, but facial movements display gaps. However, they are better than patches.
Osseointegrated implants
It is inevitable that ablation surgery will alter the movements, pliability and anatomy of the oral and facial structures. Although clever use of undercuts and interconnecting multi-pieced prostheses are effective, retention of prostheses can remain a problem.
The introduction of osseointegrated implants has been a major advancement in overcoming this problem. Placement of implants requires adequate bone stock (reconstruction with DCIA or fibula free flaps preferred to radial composite free flaps) and good patient compliance and dexterity (to maintain a high standard of hygiene) as well as clinical resources (funding, expertise and further surgery). At some sites, however, these implants can give a prosthetic reconstruction unachievable by surgical treatment alone (see Figure 11).
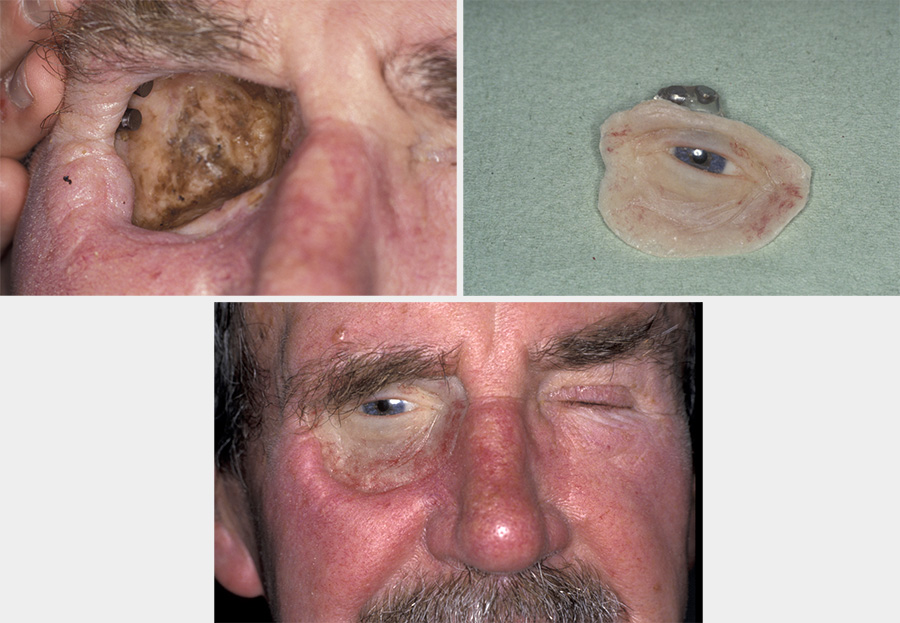
Implants can be inserted at the same time as the flap, but accurate alignment for prosthetic use is much more difficult. When the implants are inserted following adjuvant radiotherapy (the usual approach in practice), some clinicians advocate hyperbaric oxygen therapy, HBO to reduce the risk of osteoradionecrosis.
A general summary
It is important to reiterate that ablative surgery (the degree of healthy tissue sacrificed in the name of aiming to cure) must not be compromised for the sake of reconstruction. In addition, any comorbidity of a patient has a significant bearing in predicting outcomes and must be considered as part of the treatment plan.
The advantages of each reconstructive technique should be weighed, and the best possible option available (bearing in mind available expertise) to suit the patient and the defect should be used. Donor site morbidity should be taken into account.
Reconstructive surgical techniques have advanced significantly. The larger and the more complex a defect, the more impaired a patient will be functionally. Good communication is essential for properly informed decisions, for realistic expectations and for engagement in the process.
Technical innovations include preoperative virtual planning of both the resection and the reconstruction, to allow precision positioning of the osteotomies and shaping of the retaining plate with pre-planned jigs. Whether the operating time saved justifies the additional cost and (current) preoperative delay in most patients requiring osseous reconstruction (malignant disease) or whether this technology will be reserved for resections for benign disease only remains to be seen.
Ultimately, the reconstructive option chosen should be a joint decision between the surgeon and the patient, based on a balance of risks (including comorbidity) and benefits, as well as an honest assessment of the expertise available at a clinical unit. The goal must be to use the simplest and most reliable technique possible to achieve the best possible rehabilitation.