Soft tissue necrosis
Below we discuss the following three topics in a little more detail:
- how cell death by necrosis differs from the regular and ‘programmed’ cell death (apoptosis) that is an essential component of the normal body regeneration;
- how apoptosis and necrosis relate to each other;
- soft tissue necrosis conditions of particular relevance in the maxillofacial region.
Necrosis and apoptosis - processes
Both necrosis and apoptosis refer to series of events leading to cell death. The apoptosis pathway follows a genetically encoded, internal program for cell renewal and tissue regeneration. It is a healthy process in a living body and is sometimes called programmed cell death. When cells divide in the regeneration process to produce new cells (mitosis), older cells are replaced and have to be removed. This process of change goes on in the body all the time in all tissues. Apoptosis is also involved in the healing process and at various stages in the development of a foetus (for example, the fingers of a foetus develop from an early primitive form of a hand by apoptosis to separate the fingers from each other).
In contrast, the necrosis pathway is triggered by external factors such as trauma or infection and leads to damage to, and destruction of, tissues. The main mechanistic difference between the necrosis and apoptosis pathways is the absence of inflammation in the apoptosis process. The two processes are illustrated schematically in Figure 1.
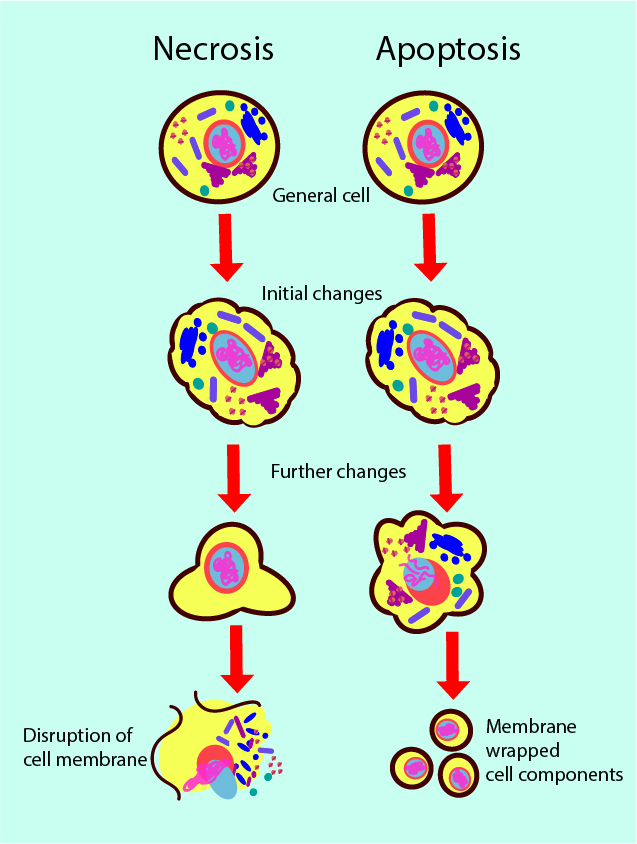
During apoptosis, a cell starts reorganising its contents and its surface in an orderly fashion. Bubble-like structures develop on the surface and the functional units (organelles) in the cell get broken down into their components. The cell starts to shrink and the broken-down units are wrapped in individual membrane layers and get released. The process releases some messenger chemicals that attract specialised white blood cells (phagocytes) which consequently engulf the broken-down cell units. Most of the components get recycled in the body, for example to produce new proteins. Similar, extremely efficient recycling loops are found everywhere in nature.
Necrosis of a cell is triggered by some external event, by injury or infection. The cell starts swelling and may form bubble-like structures on the surface, the interior cell components rapidly fail to function. Eventually the cell membrane ruptures and releases the cell’s contents to the intercellular space. This includes chemicals that harm neighbouring cells and cause inflammation. The necrosis process does not release messenger chemicals that attract phagocytes, so the necrotic debris does not get cleaned up. Accordingly, necrosis can rapidly spread through tissue (and, in fact, throughout the body). Extensive tissue damage caused by necrosis is also part of the destruction caused by sepsis.
Two mechanisms trigger the necrotic pathway: loss of energy supply to a cell (lacking blood or oxygen supply), or direct damage to the cell membrane. Depending on the cause, the manifestations of soft tissue necrosis can take different forms:
- liquefactive necrosis is typically caused by bacterial or fungal infections, the dead tissue is liquefied (‘pus’);
- coagulative necrosis is typically caused by hindered circulation (most common in the heart muscle, after an infarction);
- gaseous necrosis is a combination of liquefactive and coagulative necrosis; it is typically caused by bacteria (gas gangrene, caused by clostridium infection), some mycobacterial (tuberculosis) or fungal infections;
- fat necrosis only affects fatty tissue (most common in severe inflammation of the pancreas);
- fibrinoid necrosis blocks the arteries and is caused be deposits of fibrin (insoluble protein involved in blood clotting) and antigens / antibodies.
Necrosis can affect all tissues, soft tissues as well as hard tissues (bone). The basic principles are the same for all types of tissues, even if the life cycles and regeneration mechanisms of bones are slightly different from those of soft tissues. The most common cause for bone necrosis is failure of oxygen, glucose and water delivery at a cellular level which is most easily understood as a compromised blood supply of one form or another.
Necrosis and apoptosis – a few general remarks
Our discussion of apoptosis and necrosis has painted a binary picture of two unrelated, different processes. The reality is, as usual, less black-and-white but more nuanced. It has been said that apoptosis and necrosis are likely just the two extreme ends of a scale, where less severe injury or infection may lead to apoptosis and more severe events may trigger the necrosis pathway. For example, a minor burn will cause a small blister and heal quickly, whereas a major burn will trigger necrosis. Where exactly on such a scale cell-death events fall also depends on the host’s immune system and there are large variations between individuals.
There are further connections between the two pathways. For example, if the debris from the apoptosis processes is not removed swiftly enough, apoptosis may be followed by necrosis (post-apoptotic necrosis). Such border-crossing of the two processes is thought to be of importance in autoimmune disorders.
Subdividing apoptosis and necrosis into ‘always beneficial’ (apoptosis) and ‘always harmful’ (necrosis) categories is an oversimplification. It is true that almost always necrosis has harmful effects but there may be few special circumstances where necrosis is an overall non-harmful component of wound healing. More importantly, one should acknowledge the vast number of apoptotic events that keep living bodies in balance and regenerated every day, and that these processes are entirely beneficial and healthy (and go unnoticed in everyday life). This is true as long as the rate of apoptosis remains in harmony with its task in tissue regeneration. However, if too few or too many apoptopic processes occur, this process becomes a contributing factor in disease. Too high a rate of apoptosis is thought to play an important role in autoimmune diseases such as Parkinson’s disease or various forms of muscular dystrophy where too many nerve or muscle cells die prematurely. On the other hand, if in a community of tissue cells there are too few apoptopic events, this will usually lead to the growth of tumours, which then may turn into malignant growths (one of the key features of malignant cells is the absence of a regular apoptopic process in their cell cycle, leading to uncontrolled growth).
Soft tissue necrosis in the maxillofacial region
Oral mucosa
Necrosis of the oral mucosa is most commonly caused by infections, can develop from an abscess or from some other lesion of the oral mucosa. Necrosis of the oral mucosa as an unwanted effect of chemotherapy or especially radiotherapy in the head & neck region can occur acutely, following on from severe mucositis and ulcers during treatment periods (see Figure 2).
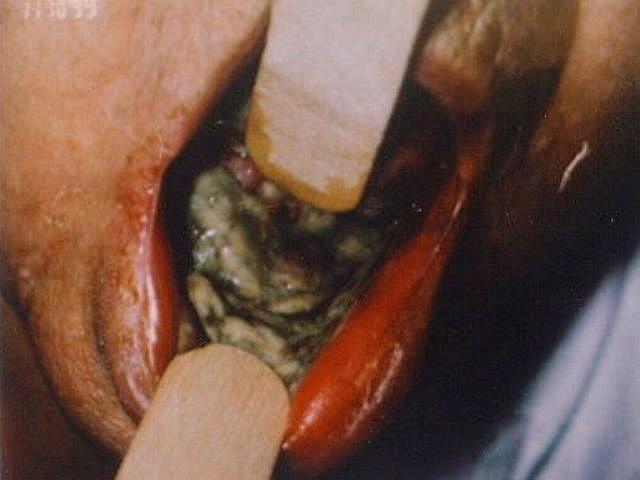
Irradiated mucosa in the mouth remains vulnerable in the long term to mechanical injury and thus remains susceptible to develop necrosis. Necrosis of the oral mucosa after radiotherapy may lead to, or occur together with, necrosis of irradiated jaw and other bones (osteoradionecrosis).
Skin
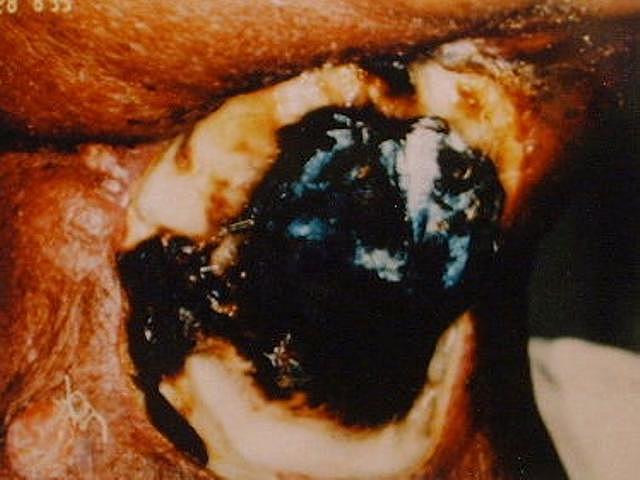
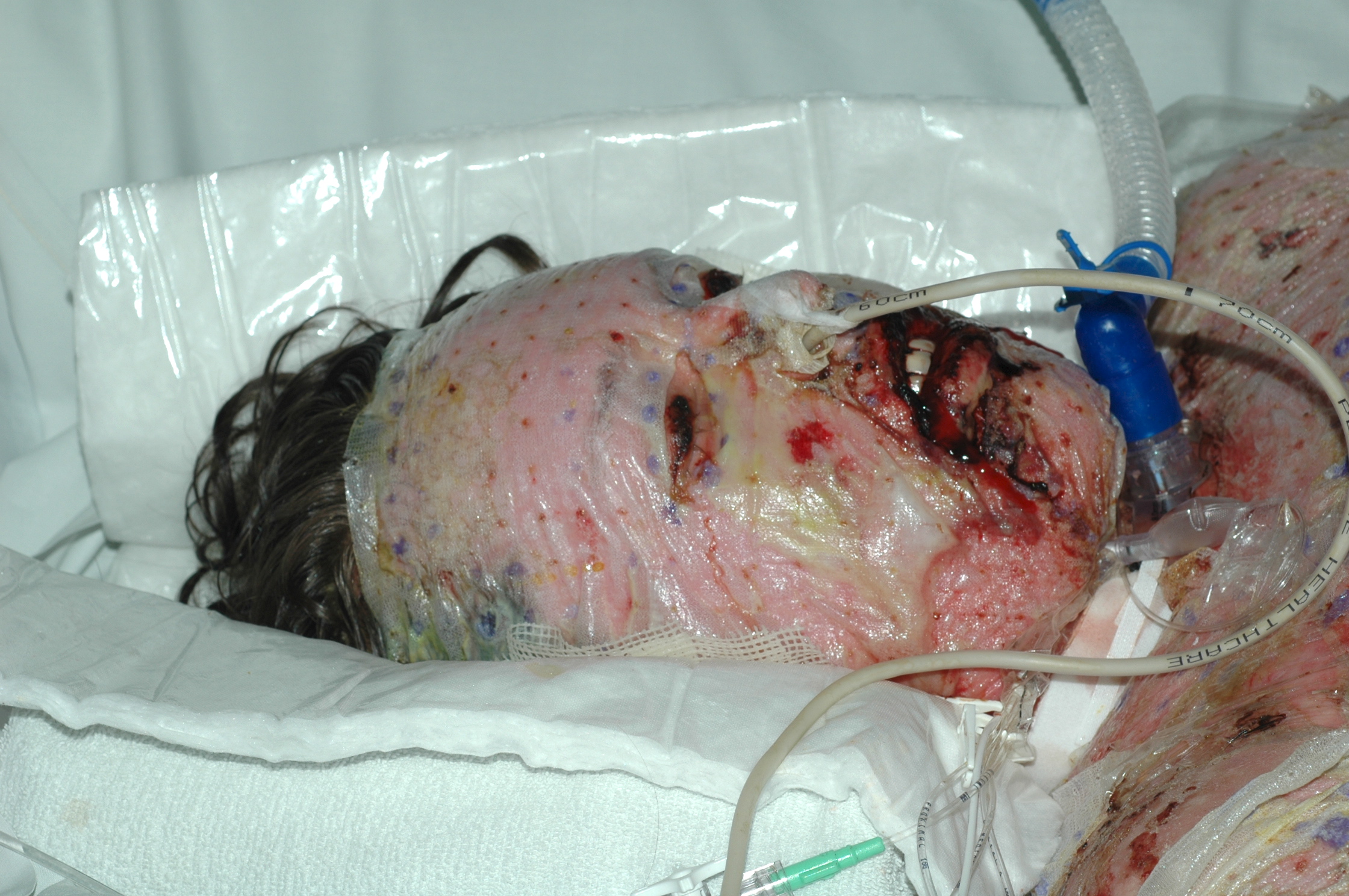
Infections of the skin, including those following trauma, in particular extended wounds and bite wounds may be the starting point for skin necrosis in the head & neck region. Skin in the head & neck region after exposure to high-energy radiation is susceptible to injury and infection and more vulnerable to necrosis than normal skin because of a typically compromised blood supply in the irradiated area.
Figure 3 and Figure 4 depict examples of skin necrosis conditions.
Deeper tissue layers (fascia, muscle)
As discussed above, the mechanisms of necrotic cell death are such that necrosis can rapidly spread through tissue. In the maxillofacial region, serious infections or abscesses can lead to necrotising fasciitis or an uncontrolled systemic inflammatory response, sepsis, both potentially life-threatening conditions.